ORIGINAL RESEARCH
Effect of endoillumination during vitrectomy on oxidative processes in rabbit blood
1 Bashkir State Medical University, Ufa, Russia
2 City Clinical Hospital No. 8, Ufa, Russia
Investigation of the mechanisms underlying retinal photodamage occurring during vitreoretinal interventions is a topical issue of ophtalmology. The study aimed to assess the effect of endoillumination of varying intensity and duration on alteration of oxidative processes in rabbit blood. The experiment involved 16 rabbits, with their retinas exposed to endoillumination of different duration (30 and 60 min) and intensity (8 and 16 cd/m2). Blood samples were collected from the rabbits’ ear vein before and after light exposure. Whole blood and serum biochemiluminescence was measured in order to assess oxidative processes. The data were analyzed using the Mann–Whitney U-test, and the results were considered significant at p ≤ 0.05. A 30-minute light exposure resulted in a significant increase in whole blood biochemiluminescence: 1.5-fold at the intensity of 8 cd/m2 and 2.5-fold at the intensity of 16 cd/m2 relative to control values (p < 0.05), indicating enhanced reactive oxygen species generation by blood cells. In contrast, a significant decrease in serum biochemiluminescence was revealed: 1.2-fold at the intensity of 8 cd/m2 and 2-fold at the intensity of 16 cd/m2 compared to control (p < 0.05) , which likely indicates a compensatory increase in antioxidant activity in response to hyperactivation of free radical processes. With the 60-minute exposure, the changes in biochemiluminescence were more pronounced: 3- and 7-fold increase in whole blood biochemiluminescence and 2- and 3-fold decrease in serum biochemiluminescence, respectively. Thus, intense light exposure resulted in the oxidative process alterations determined by the intensity and duration of exposure.
Keywords: vitreoretinal surgery, endoillumination, phototoxicity, biochemiluminescence, oxidative processes
Author contribution: Yamgutdinov RR — idea, planning the experiment, data acquisition and processing, manuscript writing and editing; Mukhamadeev TR — planning, manuscript editing; Ahmadeev RR — planning, manuscript editing; Mochalov KS — idea, manuscript writing and editing.
Compliance with ethical standards: the study was approved by the Ethics Committee of the Bashkir State Medical University (protocol No. 10 dated 11 December 2017).
Correspondence should be addressed: Rinat R. Yamgutdinov
Akademika Koroleva, 35, Ufa, 450105, Russia; moc.liamg@nirgmay
Adequate intraoperative visualization of intraocular structures accomplished using external light sources for intraocular illumination is an important prerequisite for conducting vitreoretinal surgery [1–3]. The development of new light sources for endoillumination (EI) is also associated with the transition to minor surgical approaches (25 G, 27 G, 29 G), however, the luminous flux enhancement accompanying such transition inevitably increases the risk of phototoxic damage to the retina and pigment epithelium [4, 5].
The following major parameters affecting phototoxicity of the sources of endovitreal illumination are distinguished: light source type, aphakic hazard indicator, brightness, numerical aperture of a fiber (light cone), light exposure duration, working distance (between endoilluminator and the retina), and the retinal zone affected by the luminous flux [2, 3].
Thanks decades of experimental research, the key components of pathogenesis of photodamage to the eye structures and tissues have been uncovered. Destructive photochemical reactions can occur as a result of oxidative process enhancement due to high concentrations of photosensitizers in photoreceptors, high partial pressure of oxygen in the retina and high polyunsaturated fatty acid levels [6–12]. It has been shown that the retinal pigment epithelial cells contain lipofuscin granules comprising bisretinoid fluorophores that begin to produce reactive oxygen species when exposed to light, which leads eventually to the formation of oxidized products [13].
First of all, photosensitizers, such as retinal and its metabolites, are potentially dangerous in terms of phototoxicity. These can generate singlet oxygen and superoxide radicals when exposed to light [14]. Other pathogenetic factors include high oxygen tension reaching 100 mmHg at the level of the apices of the outer photoreceptor segments. Finally, it should be noted that oxidative destruction of melanosomes can result in formation of toxic aldehydes and ketones, as well as in protein modification [15].
Thus, according to the published data, all the potentially dangerous conditions for photodamage to the eye structures and tissues, especially the photoreceptor–pigment epithelium complex, arise during vitreoretinal interventions.
The study aimed to assess the effect of endoillumination of varying intensity and duration on oxidative process alteration in rabbit blood.
METHODS
The study involving 16 rabbits (males) aged 6–9 months with the body weight of 2–3 kg was conducted at the Chair of Ophtalmology and the Central Research Laboratory of the Bashkir State Medical University. The animals were bought from specialized breeding nursery (SP Tulupov; Russia). Rabbits were kept in the controlled environment ensuring optimal temperature, humidity, and illumination; these were given a balanced diet including specialized foods and fresh water. The animals were administered 2% xylazine solution (Alfasan International B.V.; Netherlands) intramuscularly in a dose of 1 mg per 1 kg of body weight half an hour before surgery in order to ensure a pronounced sedative effect. Zoletil 100 (Virbac; France) in a dose of 7.5 mg per 1 kg of rabbit’s body weight was used intramuscularly for general anesthesia. The Mydrimax drops (Sentiss Pharma; India) were instilled into the conjunctival sac 15–20 min before the procedure to dilate pupils. Local anesthesia was achieved using the 0.5% proxymetacaine hydrochloride solution.
The test samples were represented by blood collected from the rabbits’ ear vein (EV). Such an approach was used due to the fact that we collected samples from the central retinal vein (CRV) using the 27–40 G MedOne cannula earlier, at the pilot stage of the study. However, in our experiments, blood collection directly from the CRV had serious methodological limits, since the procedure is associated with further alteration of intraocular media. The analysis of biochemiluminescence (BCL) parameters involving samples collected from the rabbits’ CRV and EV revealed no significant differences, which allowed us to use blood from EV as test samples.
A 25 G port installed in the projection of the pars plana part of the ciliary body was used to model endovitreal exposure. A xenon light source integrated into the Optimed Profi ophtalmological miscrosurgical system (Optimedservice; Russia) was used as an illuminator. The endoilluminator was introduced via the port, and its tip was placed at a distance of 8 mm (light cone 30°) from the retina in the direction of the macular zone.
The experimental animals were divided into two groups.
Group 1: exposure to light with the intensity of 8 cd/m2 (50%) for 30 min (4 rabbits, 4 eyes) and 60 min (4 rabbits, 4 eyes).
Group 2: exposure to light with the intensity of 16 cd/m2 (100%) for 30 min (4 rabbits, 4 eyes) and 60 min (4 rabbits, 4 eyes).
The follow-up collection of blood from the ear vein was performed after the end of the light exposure.
The BHL recording method was used to assess the effects of endoillumination during vitrectomy on oxidative processes. The use of BCL in vitreoretinal surgery for estimation of photodamage to the eye tissues is based on the fact that biochemiluminescence reflects light emission from the electronically excited atoms and molecules emerging during chemical reactions. Furthermore, free radical processes are the only biochemiluminescence sources [16].
We recorded blood BCL with the HLM-003 system (Ufa State Aviation Technical University; Russia) measuring the light sum S and maximum slow-flash amplitude Imax in quanta per second for 3 min [17]. The whole blood BCL was used as an indicator of the oxidative processes of reactive oxygen species generation by blood cells, primarily neutrophils. We used luminol (10-4 М) in the dimethyl sulfoxide (DMSO) solution to record luminol-dependent chemiluminescence (LDBCL). A total of 0.1 mL of blood were mixed with 2 mL of luminol solution, then the mixture was put in the chamber of the unit at 37 °С. In addition to LDBCL, we assessed iron-induced biochemiluminescence (IIBCL) of blood serum in order to determine the levels of oxidative processes of lipid peroxidation (LPO). Blood was centrifuged to obtain serum, and serum was diluted with phosphate buffer. Luminescence was indiced by adding the FeSO4×7H2O solution, the intensity of which reflected the LPO level.
The data were analyzed using the nonparametric Mann–Whitney U-test. The results were considered significant at p ≤ 0.05 and presented as median (Me) and interquartile range (IQR).
RESULTS
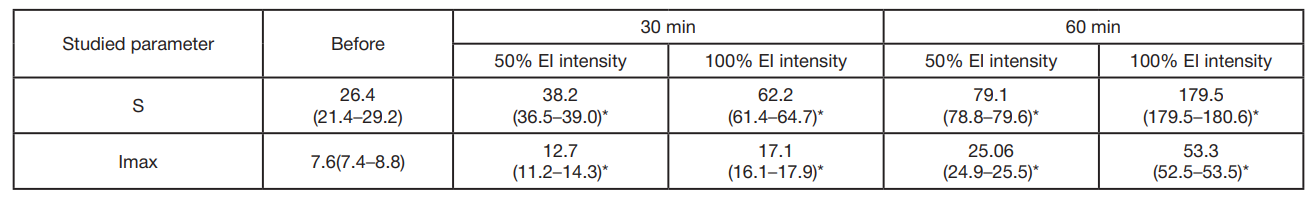
A significant increase in biochemiluminescence with luminol was observed after the 30-min exposure to light with the 50% intensity (8 cd/m2; group 1). During the experiment LDBCL increased from the baseline level of 26.4 × 105 quanta/s to 38.2 × 105 quanta/s. This increase corresponds to the almost 50% intensity growth, which suggests a significant effect of moderately intense radiation on this indicator. There was a rather more pronounced increase in LDBCL (up to 62.2 × 105 quanta/s) under exposure to radiation with greater intensity 100% (16 cd/m2; group 2). Such an increase is equivalent to the almost 2.5-fold intensity growth relative to baseline. The data obtained show that the more intense radiation has a stronger effect on LDBCL. This confirms the fact that the effect depends on the intensity of radiation used. We also conducted appropriate measurement of LDBCL for the 60-min exposure in order to assess the longer EI. The data are provided in tab. 1.
According to the table, EI leads to the LDBCL enchancement, which reflects the increased generation of free radicals by blood cells. The exposure time increase results in the LDBCL parameter enchancement, which suggests intensification of oxidative processes in blood.
The LDBCL value increased 3-fold after the 60-min exposure: the control value was 26.4 × 105 quanta/s, and the value after the exposure was 79.1 × 105 quanta/s. There was an almost 7-fold increase in LDBCL, when the intensity increased to 100% (16 cd/m2).
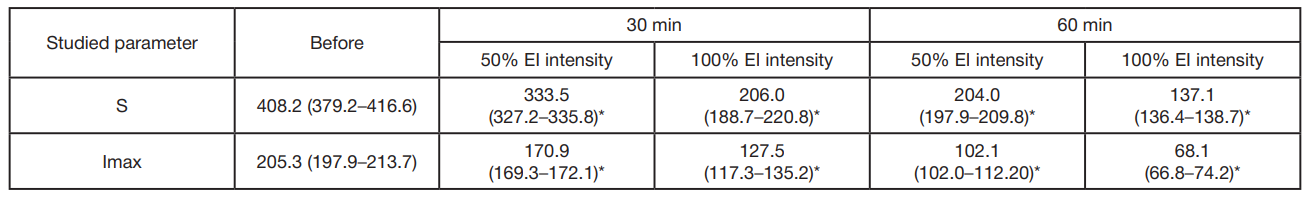
The following patterns were revealed when recording IIBCL of the samples exposed to EI: the 30-min EI source exposure resulted in the significantly decreased IIBCL parameters: 1.2-fold in the first group relative to control. The intensity increase to the full value of 100% (16 cd/m2; group 2) resulted in the 2-fold decrease in luminosity parameters. Control — 408.2 × 105 quanta/s, after the exposure — 206.1 × 105 quanta/s.
When the exposure duration increased to 60 min, the decrease in IIBCL parameters became more noticeable. At 50% intensity (8 cd/m2; group 1) IIBCL decreased to 204.0 × 105 quanta/s, which was 2 times lower. At 100% intensity (16 cd/m2; group 2) IIBCL dropped to 137.1×105 quanta/s, which was 3 times lower. These findings demonstrate that the illumination duration and intensity have a significant effect on blood serum biochemiluminescence (tab. 2).
DISCUSSION
Our findings show that alterations of oxidative processes in blood are clearly of dose-dependent nature associated with the endoillumination intensity and duration. With increasing EI intensity and duration, enhancement of LDBCL of the test samples has been reported, which suggests enhancement of oxidative processes in blood cells. It is clear that at first the oxygen-dependent cellular mechanisms ensuring realization of reactive oxygen species generation are activated.
The findings are in line with the general conclusions that oxidative processes yielding the oxidative degradation products are triggered in molecular structures of the eye under exposure to light [13, 18].
The low IIBCL values revealed reflect the antioxidant activity increase and lipid peroxidation decrease. This may be due to the fact the enhancement of oxidative processes causes activation of antioxidant enzyme systems. Certain studies show that inhibitory effect on the oxidative photodestruction products is associated with antioxidant activity [19]. The antioxidant activity increase in blood serum can probably be explained by compensatory rearrangement of the antioxidant systems caused by free radical accumulation resulting from endoillumination. This process can be considered as an adaptive mechanism allowing the body to neutralize excess free radicals and maintain cellular homeostasis.
Thus, the effects of EI exposure lead to complex adaptive response of the hemic system that is intended to reduce damage caused by free radical oxidation and maintain cell viability. The studies of mechanisms underlying light-induced damage to the eye show that the oxidative degradation products can diffuse into the cell cytoplasm and cause toxic effects even when there is no light exposure [13]. According to our study, the EI time and intensity represent an important factor of the LDBCL and IIBCL parameter alteration. The longer and more intense is EI, the more severe are alterations in the form of increased LDBCL and decreased IIBCL.
CONCLUSIONS
The data provided make it possible to conclude that the duration and intensity of exposure to light emitted by endoillumination sources lead to considerable alterations of oxidative processes in blood. The most severe alterations of oxidative processes are reported after the 60-min exposure to endoillumination of 100% intensity. Further morphological and electrophysiological testing will help determine the contribution and temporary pattern of the identified alterations of oxidation processes to the development of phototoxic risk of endoillumination sources.
- Kaziev SN, Borzenok SA, Saburina IN, Kosheleva NV, Tonaeva HD. Jendoilljuminacija v hode vitreal'noj hirurgii — jevoljucija voprosa i osobennosti primenenija na sovremennom jetape. Prakticheskaja medicina. 2013; 70: 10–2. Russian.
- de Oliveira PR, Berger AR, Chow DR. Vitreoretinal instruments: vitrectomy cutters, endoillumination and wide-angle viewing systems. Int J Retina Vitreous. 2016; 2: 28.
- McCannel CA. Advanced in endoillumination. Retinal Physician. 2015; 12: 9–10.
- Totsuka K, Ueta T, Uchida T, Roggia MF, Nakagawa S, Vavvas DG, et al. Exp Eye Res. 2019; 181: 316–24.
- Sun Y, Zheng Y, Wang C, Liu Y. Glutathione depletion induces ferroptosis, autophagy, and premature cell senescence in retinal pigment epithelial cells. Cell Death Dis. 2018; 9 (7): 753.
- Tang Z, Ju Y, Dai X, Ni N, Liu Y, Zhang D, et al. HO-1-mediated ferroptosis as a target for protection against retinal pigment epithelium degeneration. Redox Biol. 2021; (43): 101971.
- Upadhyay M, Milliner C, Bell BA, Bonilha VL. Oxidative stress in the retina and retinal pigment epithelium (RPE): Role of aging, and DJ-1. Redox Biol. 2020; 37: 101623.
- Rossino MG, Lulli M, Amato R, Cammalleri M, Monte MD et al. Oxidative Stress Induces a VEGF Autocrine Loop in the Retina: Relevance for Diabetic Retinopathy. Cells. 2020; 9 (6): 1452.
- Wang S, Ji LY, Li L, Li JM. Oxidative stress, autophagy and pyroptosis in the neovascularization of oxygeninduced retinopathy in mice. Mol Med Rep. 2019; 19 (2): 927–34.
- Ozawa Y. Oxidative stress in the light-exposed retina and its implication in age-related macular degeneration. Redox Biol. 2020; 37: 101779.
- Datta S, Cano M, Ebrahimi K, Wang L, Handa JT. The impact of oxidative stress and inflammation on RPE degeneration in non-neovascular AMD. Prog Retin Eye Res. 2017; 60: 201–18.
- Guo KX, Huang C, Wang W, Zhang P, Li Y, et al. Oxidative stress and mitochondrial dysfunction of retinal ganglion cells injury exposures in long-term blue light. Int J Ophthalmol. 2020; 13 (12): 1854–63. https://doi.org/10.18240/ijo.2020.12.03.
- Yakovleva MA, Ostrovsky DS, Khubetsova MH, Borzenok SA, Feldman TB, Ostrovsky MA. Study of the cytotoxic properties of unoxidized and oxidized bisretinoids of lipofuscin granules in retinal pigment epithelium cells. Pathological Physiology and Experimental Therapy. 2023; 67 (3): 76–87. Russian.
- Zhuravlev AI. Kvantovaja biofizika zhivotnyh i cheloveka: uchebnoe posobie. M.: Izd-vo «BINOM. Laboratorija znanij», 2015. Russian.
- Gulin AA, Dontsov AE, Yakovleva MA, Trofimova NN, Aybush AV et al. Oxidative destruction of human RPE melanosomes induced by superoxide radicals leads to the formation of reactive aldehydes and ketones. St. Petersburg State Polytechnical University Journal. Physics and Mathematics. 2022; 15 (3.2): 311–6.
- Zakoteev JuA. Hemiljuminescencija Principy i metodiki registracii, oborudovanie, zadachi. M., 2015. Russian.
- Farhutdinov RR., Tevdoradze SI. Metodiki issledovanija hemiljuminescencii biologicheskogo materiala na hemiljuminomere HL-003. Metody ocenki antioksidantnoj aktivnosti biologicheski aktivnyh veshhestv. M.: RUDN, 2005. Russian.
- Dontsov AE, Yakovleva MA, Vasin AA, Gulin AA, Aybush AV, et al. Understanding the mechanism of light-induced age-related decrease in melanin concentration in retinal pigment epithelium cells. Int J Mol Sci. 2023; 24 (17): 13099.
- Dontsov AE, Aronstam NL, Ostrovsky MA. Inhibitory effect of oxybiol on the modification of proteins by water-soluble products of photooxidative destruction of bisretinoid A2E. Biophysics. 2024; 69 (2): 257–63. Russian.