This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
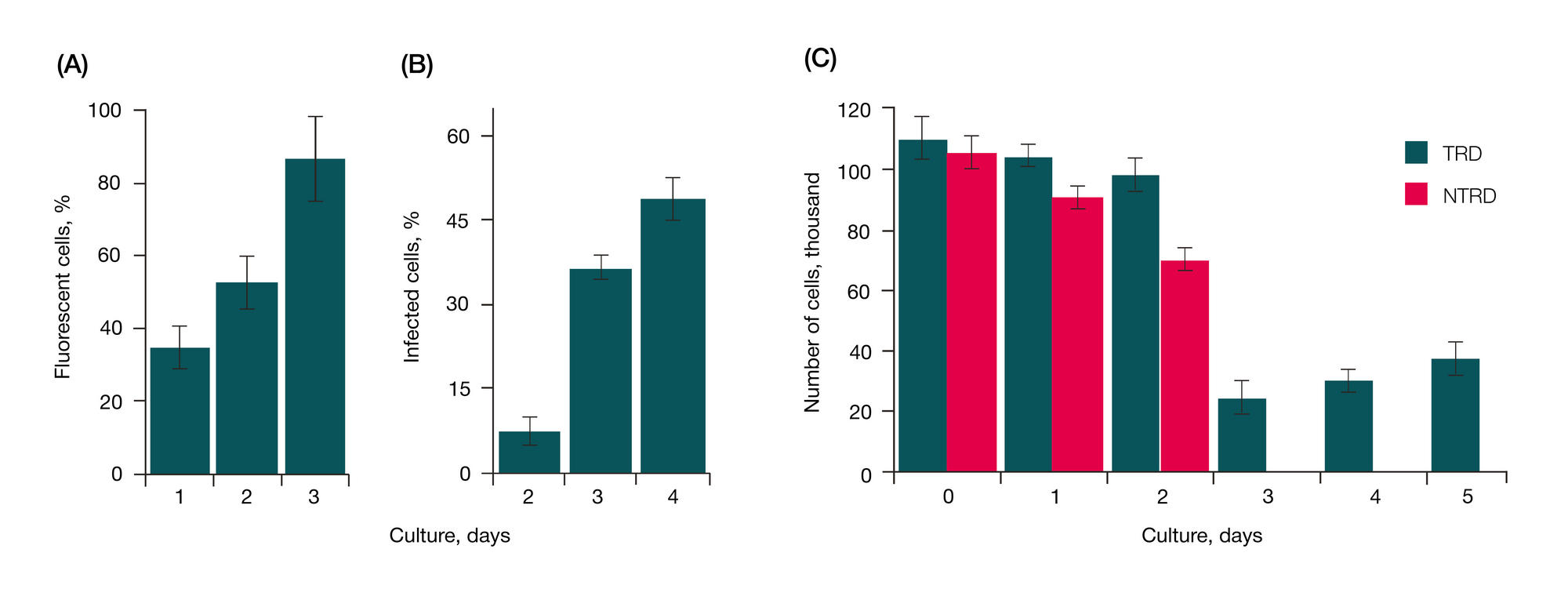
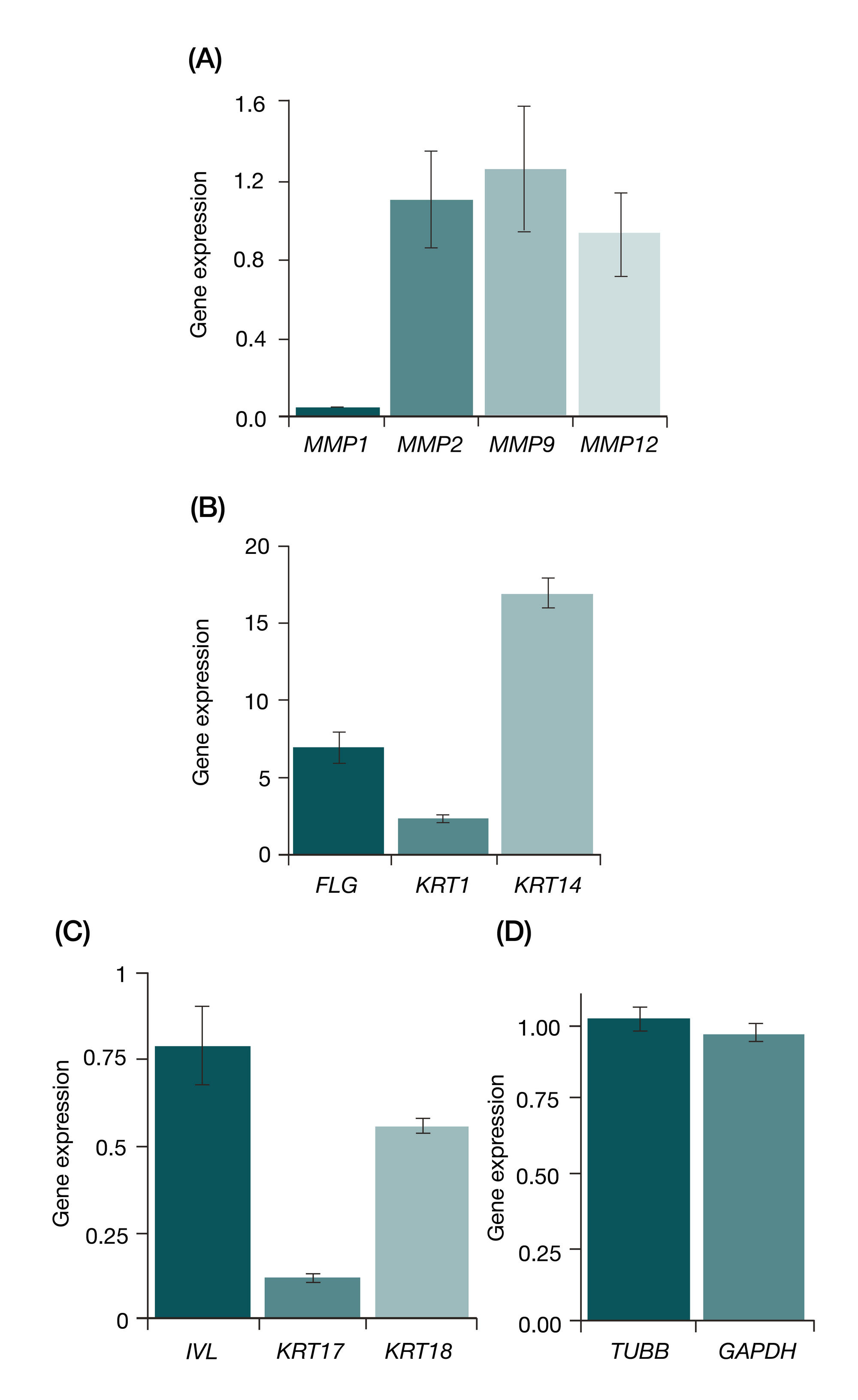
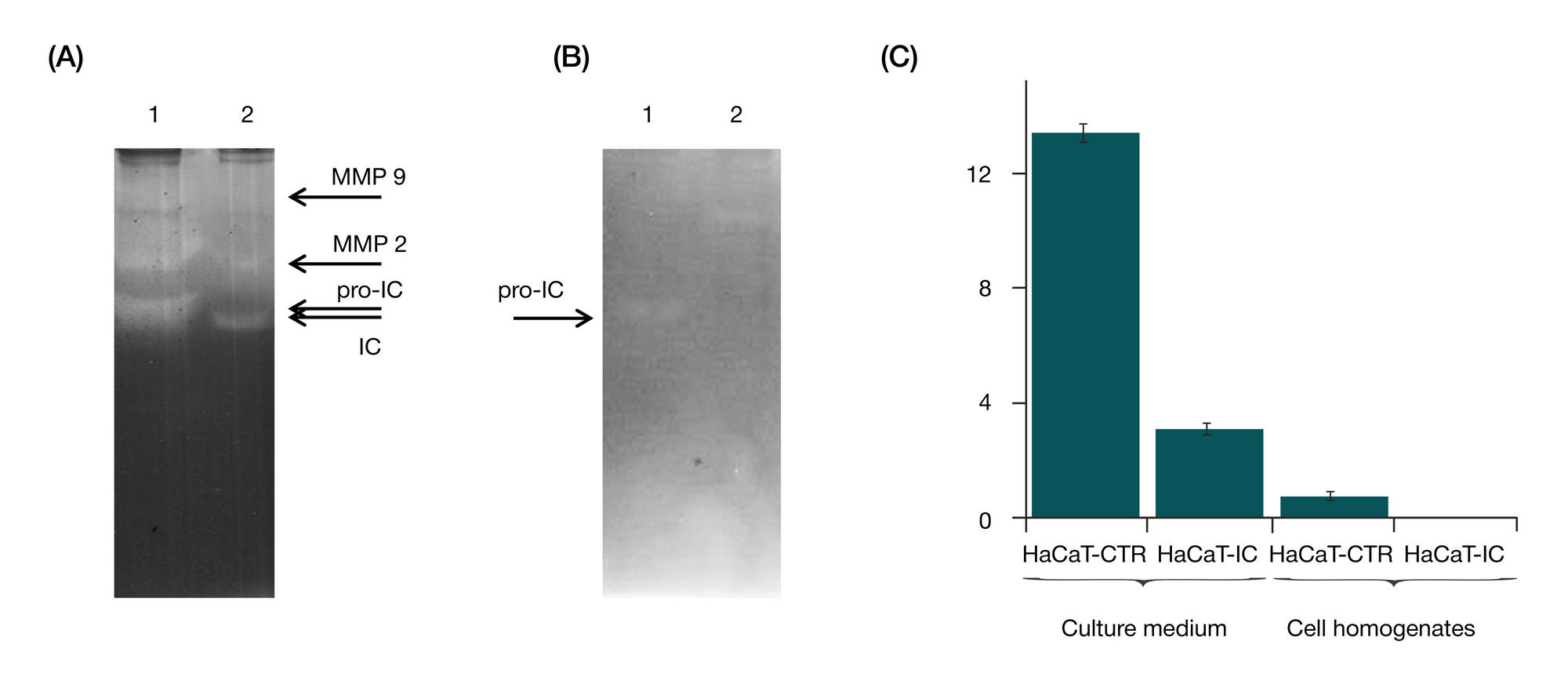
RNA interference targeting interstitial collagenase is a potential therapeutic tool to treat psoriasis
1 Russian State Agrarian University – Timiryazev Moscow Agricultural Academy, Moscow, Russia
2 Laboratory of Functional Genomics, Department of Genetics and Biotechnology,Vavilov Institute of General Genetics of the Russian Academy of Sciences, Moscow, Russia
Correspondence should be addressed: Aleksandr Mezentsev
ul. Gubkina, d. 3, Moscow, Russia, 119333; ur.ggiv@vestnesem
Acknowledgements: the authors thank Prof. Eleonora Piruzian of Vavilov Institute of General Genetics for a critical analysis of the manuscript and Prof. Maria Lagarkova of Vavilov Institute of General Genetics for the plasmids used in this study.
Contribution of all authors in data collection, analysis and interpretation was equal. Mogulevtseva AYu prepared all illustartions and Mezentsev AV drafted a manuscript. All authors participated in editing of the manuscript.