This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
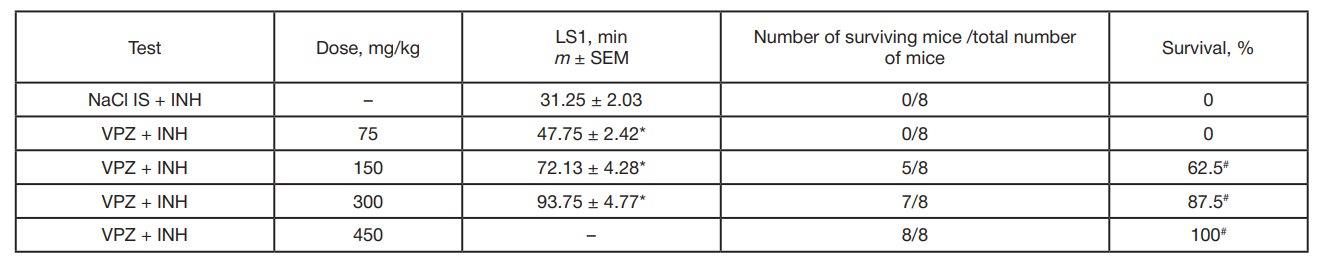
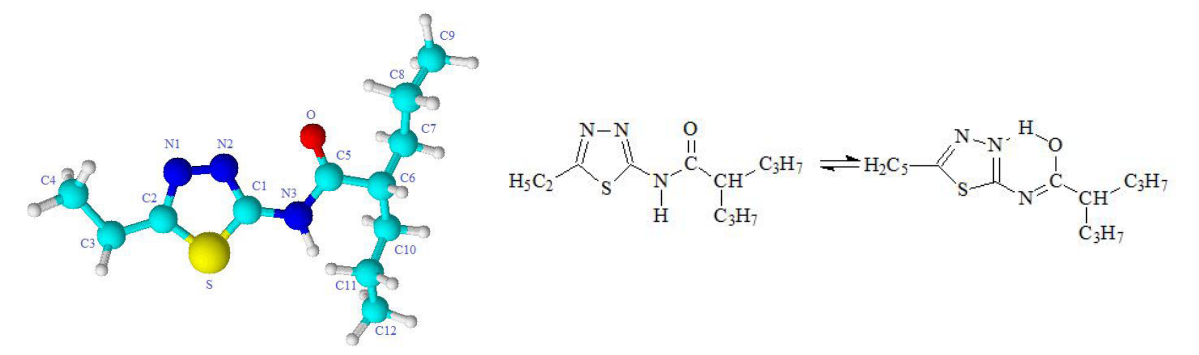
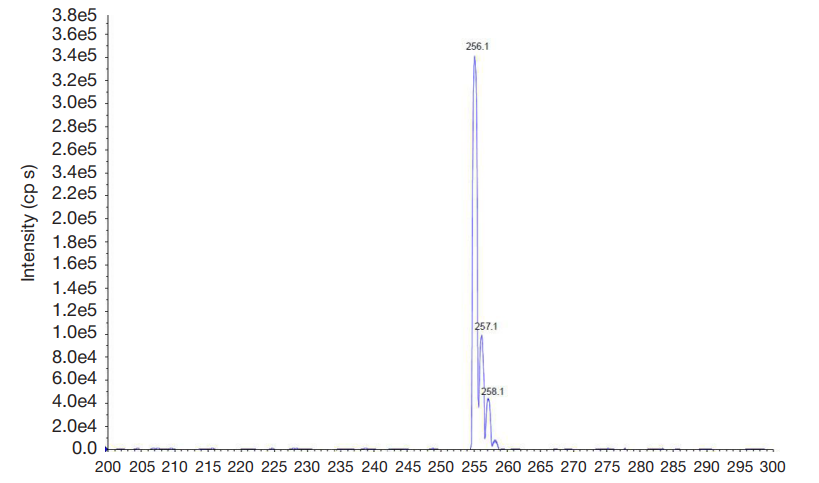
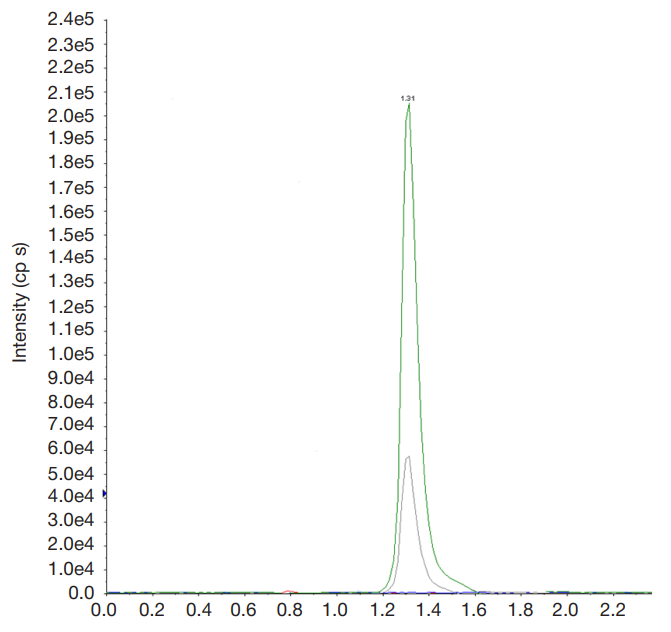
Synthesis of a novel amide derivative of valproic acid and 1,3,4-thiadiazole with antiepileptic activity
1 Tver State Medical University, Tver, Russia
2 All-Russian Research Center for the Safety of Bioactive Substances, Staraya Kupavna, Moscow region, Russia
Correspondence should be addressed: Alexandr S. Malygin
Sovetskaya, 4, Tver, 170100; ur.xednay@m.s.a.rd
Compliance with ethical standards: the study was approved by the Ethics Committee of Tver State Medical University (Protocol № 4 dated March 26, 2018). The animals were treated in compliance with the guidelines for laboratory practice in preclinical trials (Order 199n of the Russian Ministry of Healthcare dated April 1, 2016, on the Good laboratory practice). All tests were carried out in accordance with the guidelines for preclinical trials of medicinal drugs and in compliance with the European Convention for the Protection of Vertebrate Animals Used for Experimental and other Scientific Purposes (Directive 2010/63/EU).
Author contribution: Malygin AS — laboratory tests; data analysis; literature analysis; manuscript preparation; Demidova MA — study concept and design; manuscript preparation; Skachilova SYa, Shilova EV— synthesis and analysis of the compound; All authors equally contributed to the discussion of the study results.