This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
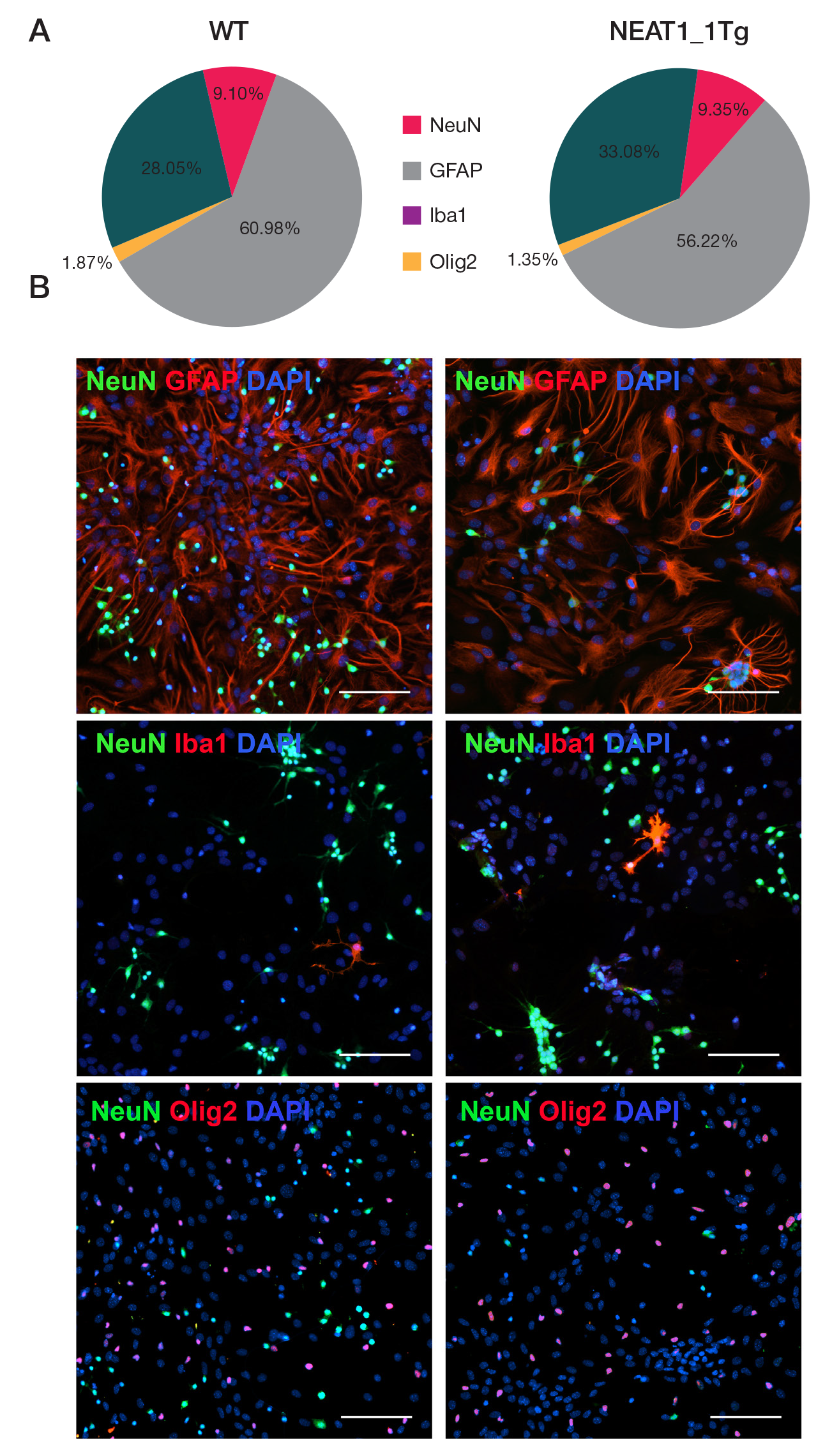
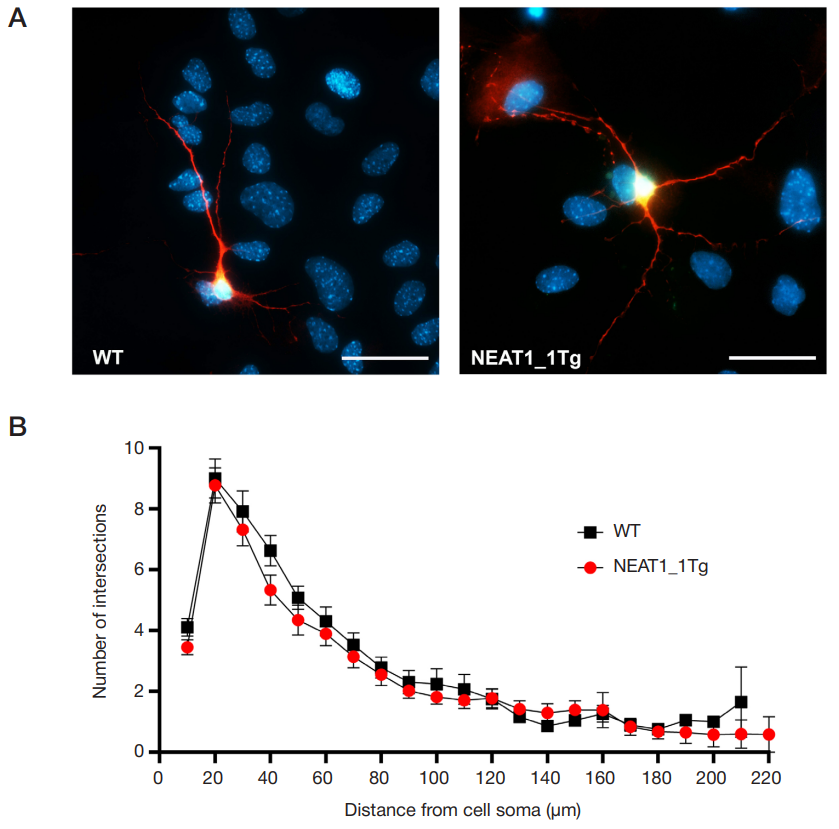
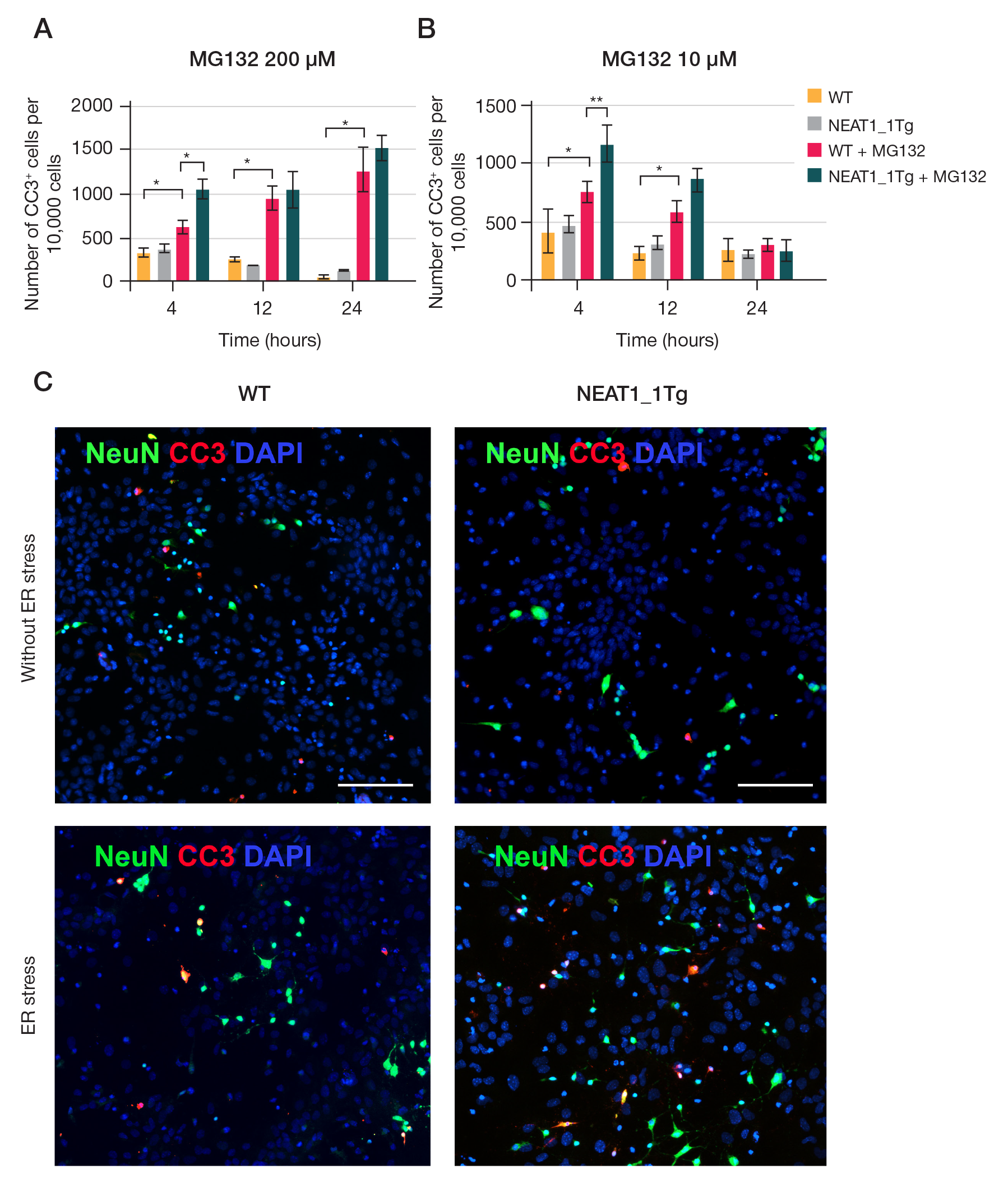
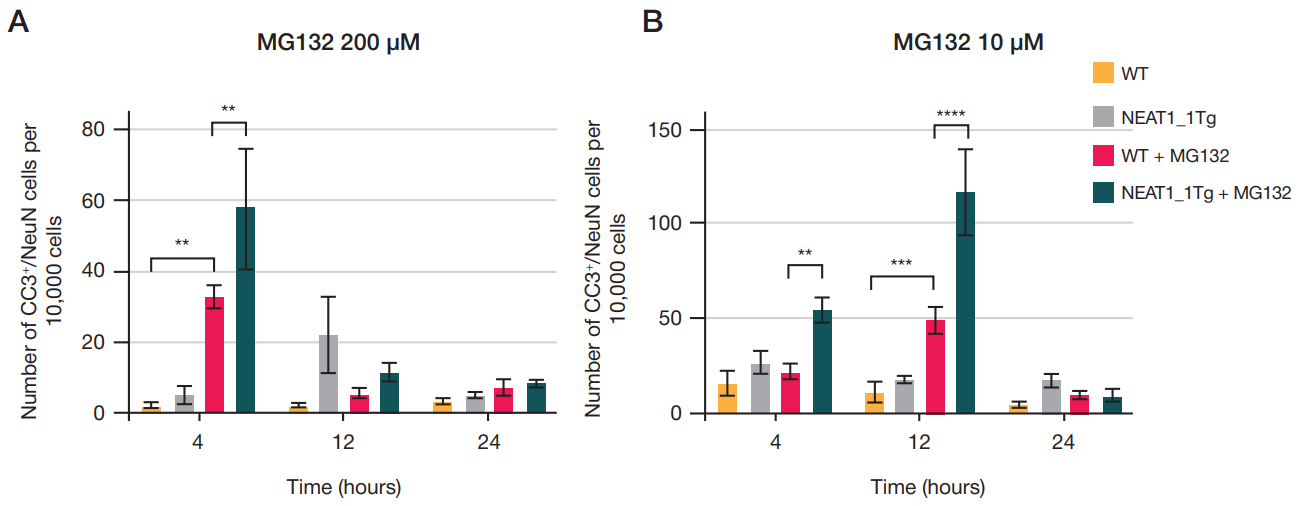
NEAT1_1 long non-coding RNA reduces the survival of primary neuronal cells under ER-stress
1 Pirogov Russian National Research Medical University, Moscow, Russia
2 Institute of Physiologically Active Compounds at Federal Research Center of Problems of Chemical Physics and Medicinal Chemistry, Russian Academy of Sciences, Chernogolovka, Russia
3 Belgorod State National Research University, Belgorod, Russia
Correspondence should be addressed: Michail S. Kukharsky
Ostrovitianova, 1/9, Moscow, 117997, Russia; ur.umsr@myksrahkuk
Funding: The study was supported by the Russian Science Foundation, Project No. 22-25-00645.
Acknowledgements: Authors would like to express their gratitude to FRC PCP MC RAS Сollective Use Center (FFSN-2021-0005 (2021 – 2023) for providing the opportunity to conduct experiments using laboratory animals.
Author contribution: Pukaeva NE — cell culture experiments, apoptosis data analysis, Zalevskaya VN — cell culture experiments, Deykin AV — generating transgenic mice, Taubinskaya MI — cell culture experiments, neuron morphology analysis, Kukharskaya OA — cell culture experiments, cell death analysis, Ovchinnikov RK — data analysis, manuscript writing, Antohin AI — manuscript editing, Kukharsky MS — study design, data analysis, manuscript writing and editing.
Compliance with ethical standards: the study was approved by the Ethics Committee of the FRC PCP MC RAS (Protocol № 53 dated December 18, 2020); animal experimental work was carried out in accordance with the Rules of Good Laboratory Practice in the Russian Federation (2016).