This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
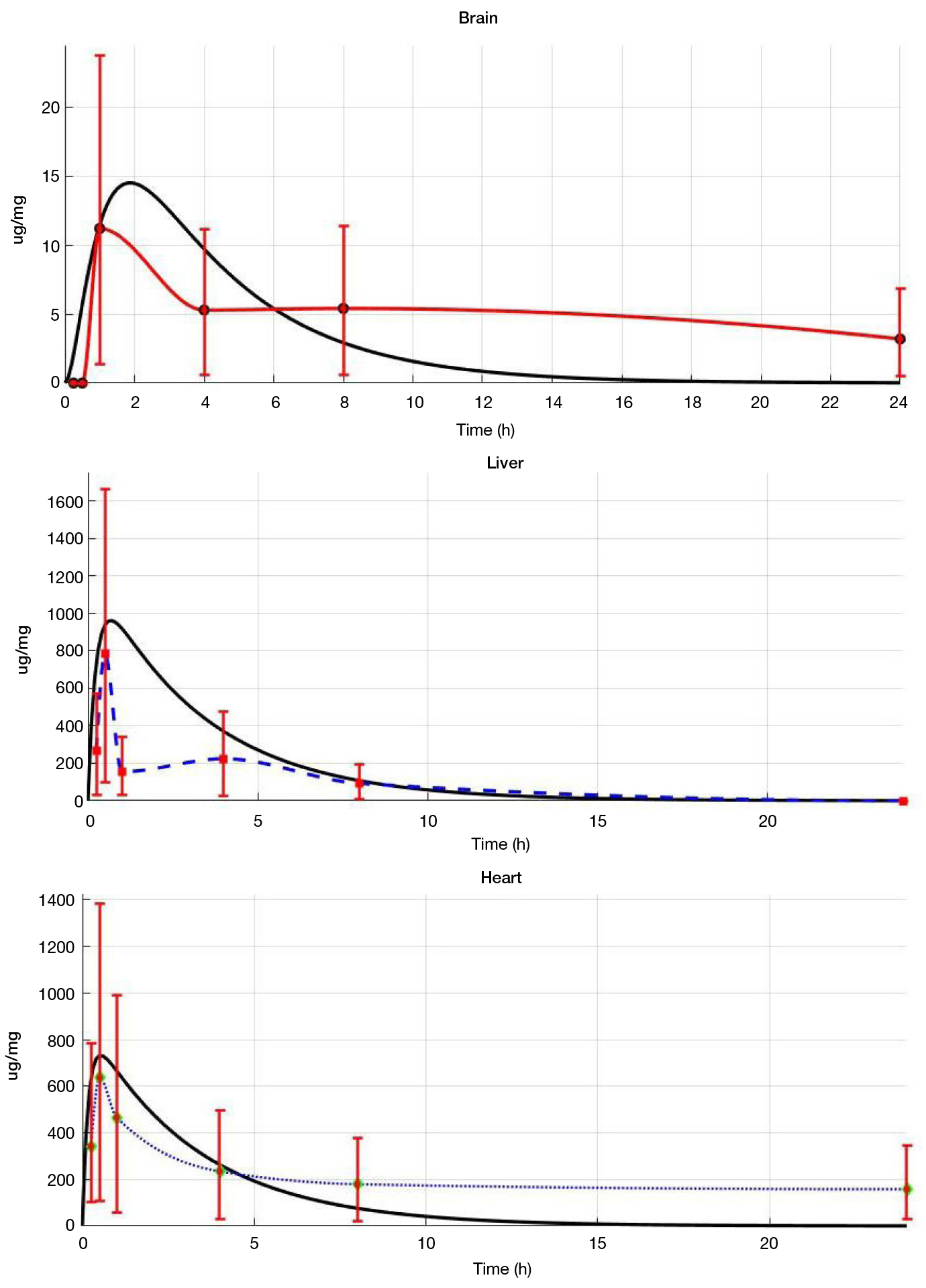
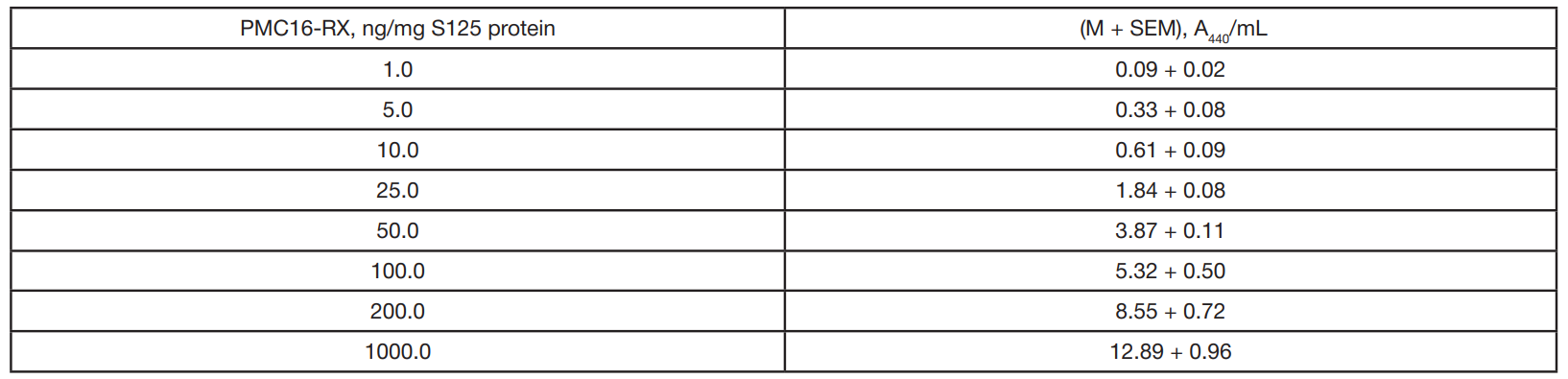
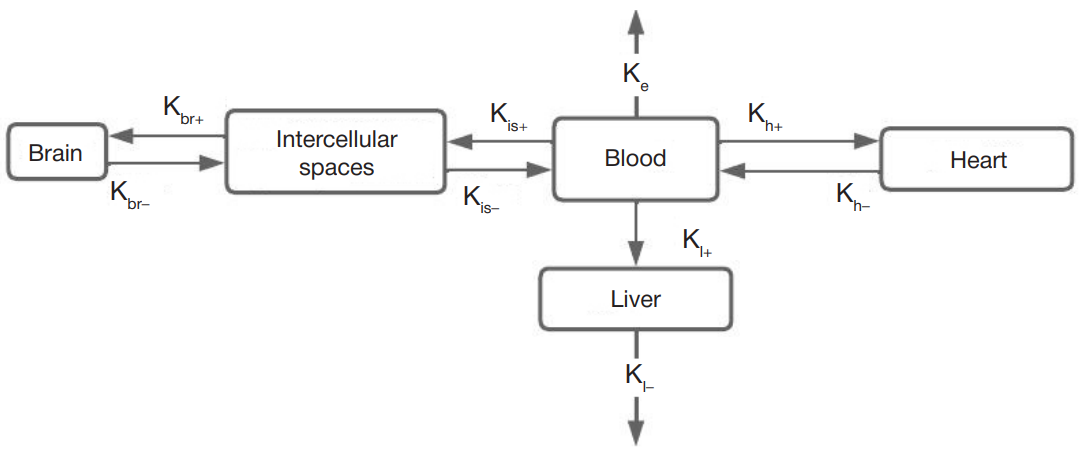
In silico algorithm for optimization of pharmacokinetic studies of [25Mg2+]porphyrin-fullerene nanoparticles
1 Mendeleev University of Chemical Technology, Moscow, Russia
2 Pirogov Russian National Research Medical University, Moscow, Russia
3 Semenov Federal Research Center for Chemical Physics, Moscow, Russia
Correspondence should be addressed: Valentin V. Fursov
Ostrovityanova, 1, Moscow, 117997, Russia; ur.liam@vosrufv
Funding: the study was funded by by the Ministry of Science and Higher Education of the Russian Federation, grant No. 075-15-2020-792 (Unique identifier RF-190220X0031)
Author contribution: Fursov VV — in silico study supervision, concept, hypothesis, structure, modeling, manuscript writing; Zinchenko DI — modeling, code, manuscript writing; Namestnikova DD — in vivo experiments; Kuznetsov DA — general supervision, data interpretation and analysis, planning of experiments.
Compliance with ethical standards: the study was approved by the ethical review board at the Pirogov Russian National Research Medical University (protocol № 140 of 15 December 2014) and the local committee for surveillance of the maintenance and use of laboratory animals (protocol № 13/2020 of 08 October 2020, protocol № 24/2021 of 10 December 2021).