This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
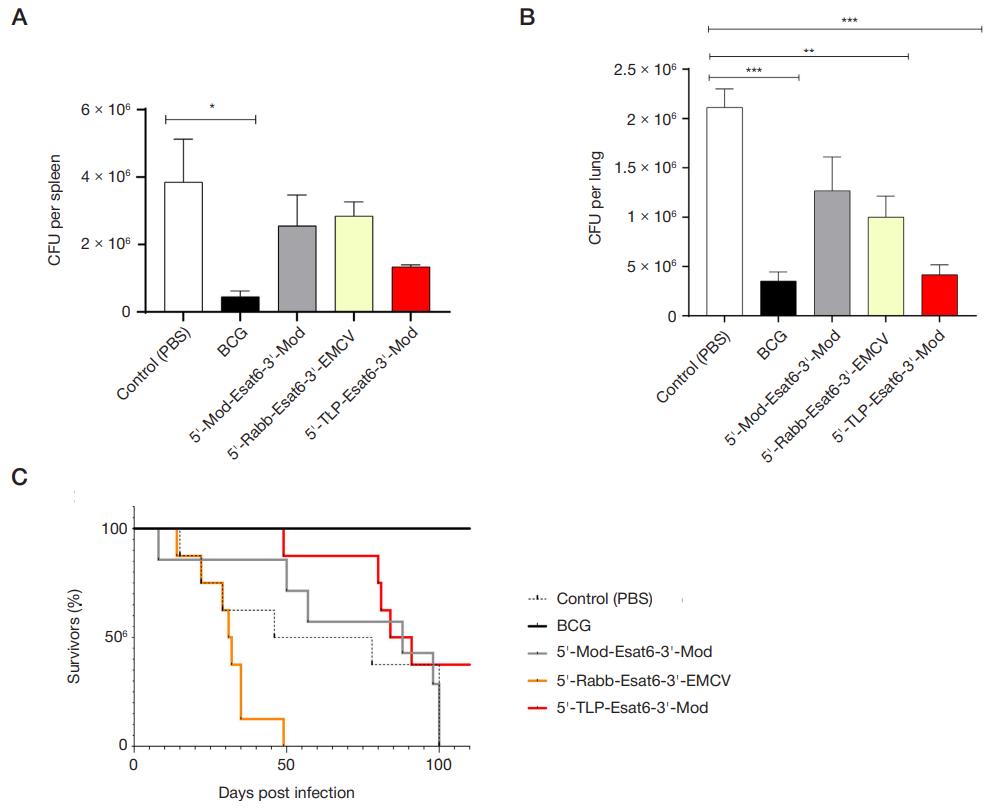
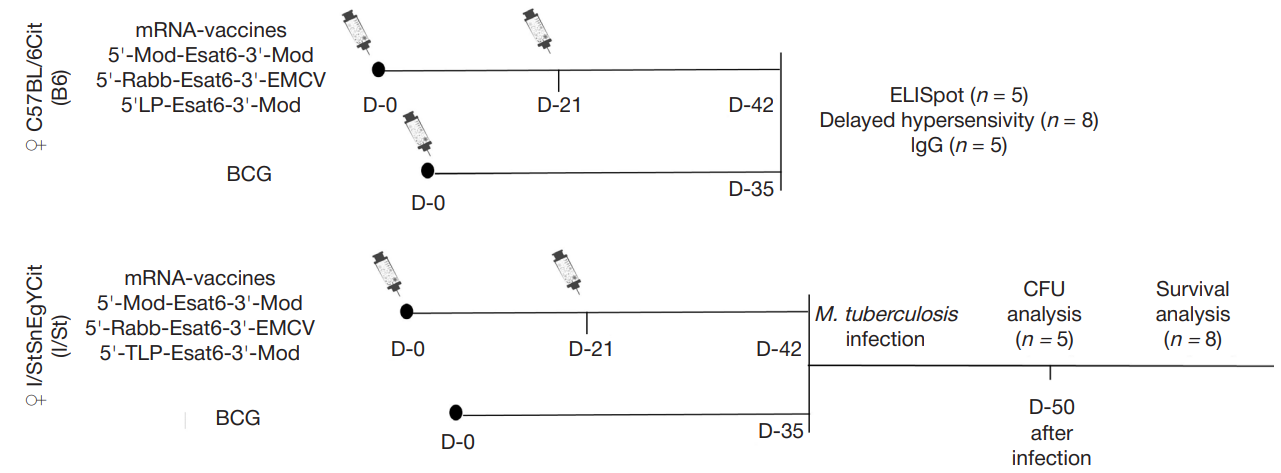
Impact of untranslated mRNA sequences on immunogenicity of mRNA vaccines against M. tuberculosis in mice
1 Central Tuberculosis Research Institute, Moscow, Russia
2 Sirius University of Science and Technology, Sochi, Russia
3 Institute of Cytology and Genetics, Siberian Branch of Russian Academy of Sciences, Novosibirsk, Russia
Correspondence should be addressed: Galina S. Shepelkova
Jauzskaja alleja, 2, 107564, Moscow, Russia; ur.irtc@avoklepehs.g
Funding: the study was supported by the Ministry of Science and Higher Education of the Russian Federation (agreement No. 075-10-2021-113, project ID RF---193021X0001).
Acknowledgements: the authors express their gratitude to staff members of the Sirius University of Science and Technology: I.M. Terenin for in vitro transcription, O.V. Zaborova for mRNA formulation into lipid nanoparticles.
Author contribution: Shepelkova GS — planning the experiments and experimental procedure (in vivo and ex vivo), data analysis, manuscript writing; Reshetnikov VV — cloning, mRNA vaccine preparation, manuscript writing; Avdienko VG — experimental procedure (in vivo and ex vivo), data analysis; Sheverev DV — mRNA vaccine preparation, data analysis; Yeremeev VV — study design, data analysis, manuscript writing; Ivanov RA — study design, manuscript writing.
Compliance with ethical standards: the study was approved by the Ethics Committee of the Central Tuberculosis Research Institute (protocol № 3/2 dated 11 May 2023) and conducted in accordance with the Order of the Ministry of Health No. 755 and the Guidelines issued by the Office of Laboratory Animal Welfare (А5502-01).